File: HAtomOrbitals.png
HAtomOrbitals.png (316 × 316 pixels, file size: 23 KB, MIME type: image/png)
Template:NowCommons
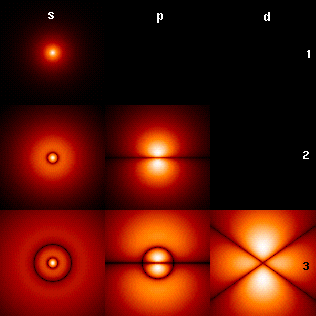
First few hydrogen atom orbitals; cross section showing color-coded probability density for different n=1,2,3 and l="s","p","d"; note: m=0
The picture shows the first few hydrogen atom orbitals (energy eigenfunctions). These are cross-sections of the probability density that are color-coded (black=zero density, white=highest density). The angular momentum quantum number l is denoted in each column, using the usual spectroscopic letter code ("s" means l=0; "p": l=1; "d": l=2). The main quantum number n (=1,2,3,...) is marked to the right of each row. For all pictures the magnetic quantum number m has been set to 0, and the cross-sectional plane is the x-z plane (z is the vertical axis). The probability density in three-dimensional space is obtained by rotating the one shown here around the z-axis.

|
Permission is granted to copy, distribute and/or modify this document under the terms of the GNU Free Documentation License, Version 1.2 or any later version published by the Free Software Foundation; with no Invariant Sections, no Front-Cover Texts, and no Back-Cover Texts. A copy of the license is included in the section entitled "Text of the GNU Free Documentation License." |
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 22:10, 25 September 2005 |  | 316 × 316 (23 KB) | David Burton (talk | contribs) | {{NowCommons|Image:HAtomOrbitals.png}} First few hydrogen atom orbitals; cross section showing color-coded probability density for different n=1,2,3 and l="s","p","d"; note: m=0 The picture shows the first few hydrogen atom [[atomic orbital | orbita |
You cannot overwrite this file.
File usage
The following 3 pages use this file: