Difference between revisions of "Template: Popular article week 51" - New World Encyclopedia
From New World Encyclopedia
Svemir Brkic (talk | contribs) m (Template:Popular article 2008 51 moved to Template:Popular article week 51: New model) |
Svemir Brkic (talk | contribs) (Added image) |
||
| Line 2: | Line 2: | ||
type=Popular| | type=Popular| | ||
title=Inner transition metal| | title=Inner transition metal| | ||
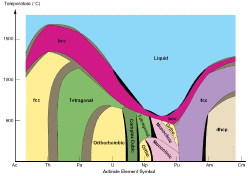
| − | image_name=| | + | image_name=Actinide_phases.gif| |
image_desc=| | image_desc=| | ||
text=The '''[[inner transition metal|inner transition metals]]''' are two series of elements known as the '''lanthanides''' and '''actinides'''. They are usually shown below all the other elements in the standard view of the [[Periodic table/standard|periodic table]], but they really belong to periods 6 and 7. The lanthanide series consists of the 14 [[chemical element|element]]s, cerium through lutetium (atomic numbers 58–71) immediately following [[lanthanum]]. Likewise, the actinide series consists of the 14 elements, thorium through lawrencium (atomic numbers 90–103), immediately following [[actinium]]. These elements were among the last to be discovered and placed in the periodic table. Many of the actinides do not occur naturally but were synthesized through nuclear reactions. | text=The '''[[inner transition metal|inner transition metals]]''' are two series of elements known as the '''lanthanides''' and '''actinides'''. They are usually shown below all the other elements in the standard view of the [[Periodic table/standard|periodic table]], but they really belong to periods 6 and 7. The lanthanide series consists of the 14 [[chemical element|element]]s, cerium through lutetium (atomic numbers 58–71) immediately following [[lanthanum]]. Likewise, the actinide series consists of the 14 elements, thorium through lawrencium (atomic numbers 90–103), immediately following [[actinium]]. These elements were among the last to be discovered and placed in the periodic table. Many of the actinides do not occur naturally but were synthesized through nuclear reactions. | ||
Chemically, the elements within each series (especially the lanthanides) are very similar to one another.}} | Chemically, the elements within each series (especially the lanthanides) are very similar to one another.}} | ||
Latest revision as of 05:08, 7 December 2009
Popular Article: Inner transition metal
The inner transition metals are two series of elements known as the lanthanides and actinides. They are usually shown below all the other elements in the standard view of the periodic table, but they really belong to periods 6 and 7. The lanthanide series consists of the 14 elements, cerium through lutetium (atomic numbers 58–71) immediately following lanthanum. Likewise, the actinide series consists of the 14 elements, thorium through lawrencium (atomic numbers 90–103), immediately following actinium. These elements were among the last to be discovered and placed in the periodic table. Many of the actinides do not occur naturally but were synthesized through nuclear reactions.
Chemically, the elements within each series (especially the lanthanides) are very similar to one another.