Difference between revisions of "Cortisone" - New World Encyclopedia
Rick Swarts (talk | contribs) |
Rick Swarts (talk | contribs) |
||
| Line 32: | Line 32: | ||
'''Cortisone''' (17-hydroxy-11-dehydrocorticosterone) is a naturally occuring [[steroid]] [[hormone]] that functions in carbohydrate metabolism and is used medically for the treatment of various ailments, including rheumatoid arthritis and certain allergies. Its formula is formula C<sub>21</sub>H<sub>28</sub>O<sub>5</sub> and IUPAC name is 17,21-dihydroxypregn-4-ene-3,11,20-trione. | '''Cortisone''' (17-hydroxy-11-dehydrocorticosterone) is a naturally occuring [[steroid]] [[hormone]] that functions in carbohydrate metabolism and is used medically for the treatment of various ailments, including rheumatoid arthritis and certain allergies. Its formula is formula C<sub>21</sub>H<sub>28</sub>O<sub>5</sub> and IUPAC name is 17,21-dihydroxypregn-4-ene-3,11,20-trione. | ||
| − | Cortisone is a [[corticosteroid]], a term that refers to steroid hormones that are produced in the adrenal cortex. Cortisone and | + | Cortisone is a [[corticosteroid]], a term that refers to steroid hormones that are produced in the adrenal cortex. Cortisone is essential for life and failure for the adrenal glands to produce it is fatal unless replacement cortisone is given. |
| − | Cortisone is | + | ==Overview== |
| + | |||
| + | Cortisone is synthesized from [[cholesterol]] in the adrenal cortex via the stimulation of adrenocorticotropin hormone (ACTH) (Gramene 2007). | ||
| + | |||
| + | Cortisone is the inactive precursor molecule of the active hormone [[cortisol]], the "stress hormone." It is activated through [[hydroxylation]] of the 11-keto-group by an [[enzyme]] called 11-beta-steroid dehydrogenase. The active form cortisol is thus sometimes referred to as [[hydrocortisone]]. | ||
| − | + | Both cortisone and cortisol are classified as glucocorticoids, a group of corticosteroids that controls protein, fat, carbohydrate, and calcium metabolism. (Minealocorticoids, the other group of corticosteroids, regulates salt and potassium levels and water retention.) Another hormone produced in the [[adrenal gland]]s, albeit in the adrenal medulla, not the adrenal cortex, is adrenaline (epinephrine), which like cortison deals with stress. | |
| − | + | Cortisone is involved in carbohydrate metablolism, with such effects listed as increased glucose release from the liver, increased liver glycogen synthesis, and decreased utilization of glucose by the tissues (Gramene 2007). | |
| − | |||
| − | + | ==Medical use== | |
Cortisone is sometimes used as a drug to treat a variety of ailments. It can be administered [[intravenous]]ly or [[cutaneous]]ly. | Cortisone is sometimes used as a drug to treat a variety of ailments. It can be administered [[intravenous]]ly or [[cutaneous]]ly. | ||
| Line 51: | Line 54: | ||
Cortisone was first discovered by the American chemist [[Edward Calvin Kendall]]. He won the 1950 [[Nobel Prize for Physiology or Medicine]] along with [[Philip S. Hench]] and [[Tadeus Reichstein]] for the discovery of [[adrenal cortex]] hormones, their structures, and functions. | Cortisone was first discovered by the American chemist [[Edward Calvin Kendall]]. He won the 1950 [[Nobel Prize for Physiology or Medicine]] along with [[Philip S. Hench]] and [[Tadeus Reichstein]] for the discovery of [[adrenal cortex]] hormones, their structures, and functions. | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
==References== | ==References== | ||
| Line 71: | Line 79: | ||
| volume=10 }} | | volume=10 }} | ||
| − | == | + | Gramene. 2007. [http://www.gramene.org/db/ontology/search?query=carbohydrate%20metabolism&ontology_type=GO Summary for carbohydrate metabolism]. |
| − | |||
| − | |||
| − | |||
| − | |||
{{credit|100857154}} | {{credit|100857154}} | ||
[[Category:Life sciences]] | [[Category:Life sciences]] | ||
Revision as of 03:52, 10 February 2007
| Cortisone | |
|---|---|
| |
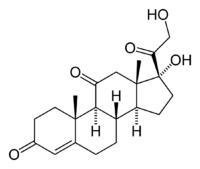
| Systematic name | 17,21-dihydroxypregn-4-ene-3,11,20-trione |
| Chemical formula | C21H28O5 |
| Molecular mass | 360.46 g/mol |
| Density | ? g/cm3 |
| Melting point | 220-224 °C |
| CAS number | [53-06-5] |
| SMILES | C[C@@](C3)4[C@](CC[C@@](O)4 [C@@](CO)=O)([H])[C@]2([H])CCC1=CC (CC[C@@](C)1[C@]([H])2C3=O)=O |
| Disclaimer and references | |
Cortisone (17-hydroxy-11-dehydrocorticosterone) is a naturally occuring steroid hormone that functions in carbohydrate metabolism and is used medically for the treatment of various ailments, including rheumatoid arthritis and certain allergies. Its formula is formula C21H28O5 and IUPAC name is 17,21-dihydroxypregn-4-ene-3,11,20-trione.
Cortisone is a corticosteroid, a term that refers to steroid hormones that are produced in the adrenal cortex. Cortisone is essential for life and failure for the adrenal glands to produce it is fatal unless replacement cortisone is given.
Overview
Cortisone is synthesized from cholesterol in the adrenal cortex via the stimulation of adrenocorticotropin hormone (ACTH) (Gramene 2007).
Cortisone is the inactive precursor molecule of the active hormone cortisol, the "stress hormone." It is activated through hydroxylation of the 11-keto-group by an enzyme called 11-beta-steroid dehydrogenase. The active form cortisol is thus sometimes referred to as hydrocortisone.
Both cortisone and cortisol are classified as glucocorticoids, a group of corticosteroids that controls protein, fat, carbohydrate, and calcium metabolism. (Minealocorticoids, the other group of corticosteroids, regulates salt and potassium levels and water retention.) Another hormone produced in the adrenal glands, albeit in the adrenal medulla, not the adrenal cortex, is adrenaline (epinephrine), which like cortison deals with stress.
Cortisone is involved in carbohydrate metablolism, with such effects listed as increased glucose release from the liver, increased liver glycogen synthesis, and decreased utilization of glucose by the tissues (Gramene 2007).
Medical use
Cortisone is sometimes used as a drug to treat a variety of ailments. It can be administered intravenously or cutaneously.
One of cortisone's effects on the body, and a potentially harmful side effect when administered clinically, is the suppression of the immune system. This is an explanation for the apparent correlation between high stress and sickness.
Cortisone is less important than a similar steroid cortisol. Cortisol is responsible for 95% of the effects of the glucocorticosteroids while cortisone is about 4 or 5%. Corticosterone is even less important.
Cortisone was first discovered by the American chemist Edward Calvin Kendall. He won the 1950 Nobel Prize for Physiology or Medicine along with Philip S. Hench and Tadeus Reichstein for the discovery of adrenal cortex hormones, their structures, and functions.
ReferencesISBN links support NWE through referral fees
- Merck Index, 11th Edition, 2533
- Woodward R. B., Sondheimer F., Taub D. (1951). The Total Synthesis of Cortisone. Journal of the American Chemical Society 73: 4057 - 4057.
- Ingle D. J. (1950). The biologic properties of cortisone: a review. Journal of Clinical Endocrinology 10: 1312-1354.
Gramene. 2007. Summary for carbohydrate metabolism.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.