Difference between revisions of "Functional group" - New World Encyclopedia
(added credit and category tags, deleted foreign language links) |
|||
| (10 intermediate revisions by 5 users not shown) | |||
| Line 1: | Line 1: | ||
| − | {{ | + | {{Copyedited}}{{Images OK}}{{Submitted}}{{Approved}}{{Paid}} |
| − | In [[organic chemistry]], '''functional groups''' (or moieties) are specific groups of [[atom]]s within [[molecule]]s, that | + | In [[organic chemistry]], '''functional groups''' (or moieties) are specific groups of [[atom]]s within [[molecule]]s, that are responsible for the characteristic [[chemical reaction]]s of those molecules. The same functional group will undergo the same or similar chemical reaction(s) regardless of the size of the molecule it is a part of. |
Combining the names of functional groups with the names of the parent [[alkane]]s generates a powerful [[systematic name|systematic nomenclature]] for naming [[organic compound]]s. | Combining the names of functional groups with the names of the parent [[alkane]]s generates a powerful [[systematic name|systematic nomenclature]] for naming [[organic compound]]s. | ||
| − | + | {{toc}} | |
| − | The non-hydrogen atoms of functional groups are always associated with each other and with the rest of the molecule by [[covalent bond]]s. When the group of atoms is associated with the rest of the molecule primarily by ionic forces, the group is referred to more properly as a [[polyatomic ion]] or [[complex ion]] | + | The non-[[hydrogen]] atoms of functional groups are always associated with each other and with the rest of the molecule by [[covalent bond]]s. When the group of atoms is associated with the rest of the molecule primarily by ionic forces, the group is referred to more properly as a [[polyatomic ion]] or [[complex ion]]—all of these are called [[radical (chemistry)|radical]]s, by a meaning of the term ''radical'' that predates the [[free radical]]. |
The first [[carbon]] atom after the carbon that attaches to the functional group is called the [[alpha carbon]]. | The first [[carbon]] atom after the carbon that attaches to the functional group is called the [[alpha carbon]]. | ||
| − | Functional groups are attached to the carbon backbone of organic molecules. | + | Functional groups are attached to the carbon backbone of organic molecules. They determine the characteristics and chemical reactivity of molecules. Functional groups are far less stable than the carbon backbone and are likely to participate in chemical reactions. |
== Table of common functional groups == | == Table of common functional groups == | ||
| Line 36: | Line 36: | ||
| [[Image:Alcohol-(general)-skeletal.png|75px|Hydroxyl]] | | [[Image:Alcohol-(general)-skeletal.png|75px|Hydroxyl]] | ||
| hydroxy- || -ol | | hydroxy- || -ol | ||
| − | | [[Image:Methanol-2D.png|90px|methanol]]<br>[[Methanol]] | + | | [[Image:Methanol-2D.png|90px|methanol]]<br/>[[Methanol]] |
|- | |- | ||
| − | | [[Aldehyde]] || | + | | [[Aldehyde]] || Aldehyde |
| RCHO | | RCHO | ||
| [[Image:Aldehyde-skeletal.png|75px|Aldehyde]] | | [[Image:Aldehyde-skeletal.png|75px|Aldehyde]] | ||
| aldo- || -al | | aldo- || -al | ||
| − | | [[Image:Acetaldehyde-skeletal.png|75px|acetaldehyde]]<br>[[Acetaldehyde]]<br/>''(Ethanal)'' | + | | [[Image:Acetaldehyde-skeletal.png|75px|acetaldehyde]]<br/>[[Acetaldehyde]]<br/>''(Ethanal)'' |
|- | |- | ||
| [[Alkane]] || [[Alkyl]] | | [[Alkane]] || [[Alkyl]] | ||
| Line 48: | Line 48: | ||
| [[Image:Alkyl-(general)-skeletal.png|75px|Alkyl]] | | [[Image:Alkyl-(general)-skeletal.png|75px|Alkyl]] | ||
| alkyl- || -ane | | alkyl- || -ane | ||
| − | | [[Image:Methane-2D-stereo.svg|75px|methane]]<br>[[Methane]] | + | | [[Image:Methane-2D-stereo.svg|75px|methane]]<br/>[[Methane]] |
|- | |- | ||
| − | | [[Alkene]] | | + | | [[Alkene]] || Alkenyl |
| R<sub>2</sub>C=CR<sub>2</sub> | | R<sub>2</sub>C=CR<sub>2</sub> | ||
| [[Image:Alkene-(general)-skeletal.png|75px|Alkene]] | | [[Image:Alkene-(general)-skeletal.png|75px|Alkene]] | ||
| alkenyl- || -ene | | alkenyl- || -ene | ||
| − | | [[Image:Ethylene.svg|75px|ethylene]]<br>[[Ethylene]]<br/>''(Ethene)'' | + | | [[Image:Ethylene.svg|75px|ethylene]]<br/>[[Ethylene]]<br/>''(Ethene)'' |
|- | |- | ||
| − | | [[Alkyne]] | | + | | [[Alkyne]] || Alkynyl |
| RC≡CR' | | RC≡CR' | ||
| [[Image:Alkyne-(general)-skeletal.png|100px|Alkyne]] | | [[Image:Alkyne-(general)-skeletal.png|100px|Alkyne]] | ||
| alkynyl- || -yne | | alkynyl- || -yne | ||
| − | |||
|- | |- | ||
| [[Amide]] || [[Carboxamide]] | | [[Amide]] || [[Carboxamide]] | ||
| Line 66: | Line 65: | ||
| [[Image:Amide-(tertiary)-skeletal.png|75px|Amide]] | | [[Image:Amide-(tertiary)-skeletal.png|75px|Amide]] | ||
| carboxamido- || -amide | | carboxamido- || -amide | ||
| − | |||
|- | |- | ||
| rowspan="4" | [[Amine]]s | | rowspan="4" | [[Amine]]s | ||
| Line 73: | Line 71: | ||
| [[Image:1°-amino-group.png|75px|Primary amine]] | | [[Image:1°-amino-group.png|75px|Primary amine]] | ||
| amino- || -amine | | amino- || -amine | ||
| − | | [[Image:Methylamine-2D.png|75px|methylamine]]<br>[[Methylamine]]<br/>''(Methanamine)'' | + | | [[Image:Methylamine-2D.png|75px|methylamine]]<br/>[[Methylamine]]<br/>''(Methanamine)'' |
|- | |- | ||
| [[amine|Secondary amine]] | | [[amine|Secondary amine]] | ||
| Line 79: | Line 77: | ||
| [[Image:Amine-(secondary).png|75px|Secondary amine]] | | [[Image:Amine-(secondary).png|75px|Secondary amine]] | ||
| amino- || -amine | | amino- || -amine | ||
| − | | [[Image:Dimethylamine-2D.png|75px|dimethylamine]]<br>[[Dimethylamine]] | + | | [[Image:Dimethylamine-2D.png|75px|dimethylamine]]<br/>[[Dimethylamine]] |
|- | |- | ||
| [[amine|Tertiary amine]] | | [[amine|Tertiary amine]] | ||
| Line 85: | Line 83: | ||
| [[Image:Amine-(tertiary).png|75px|Tertiary amine]] | | [[Image:Amine-(tertiary).png|75px|Tertiary amine]] | ||
| amino- || -amine | | amino- || -amine | ||
| − | | [[Image:Trimethylamine chemical structure.png|75px|trimethylamine]]<br>[[Trimethylamine]] | + | | [[Image:Trimethylamine chemical structure.png|75px|trimethylamine]]<br/>[[Trimethylamine]] |
|- | |- | ||
| [[Quaternary ammonium cation|4° ammonium ion]] | | [[Quaternary ammonium cation|4° ammonium ion]] | ||
| Line 91: | Line 89: | ||
| [[Image:Quaternary-ammonium-cation.svg|75px|Quaternary ammonium cation]] | | [[Image:Quaternary-ammonium-cation.svg|75px|Quaternary ammonium cation]] | ||
| ammonio- || -ammonium | | ammonio- || -ammonium | ||
| − | | [[Image:Choline-skeletal.png|150px|choline]]<br>[[Choline]] | + | | [[Image:Choline-skeletal.png|150px|choline]]<br/>[[Choline]] |
|- | |- | ||
| [[Azide]] | | [[Azide]] | ||
| Line 112: | Line 110: | ||
| [[Image:Benzyl-group.png|75px|Benzyl]] | | [[Image:Benzyl-group.png|75px|Benzyl]] | ||
| benzyl- | | benzyl- | ||
| − | | 1- | + | | 1-''(substituent)''toluene |
| [[Image:Benzyl-bromide-skeletal.svg|75px|Benzyl bromide]]<br />[[Benzyl bromide]]<br />''(1-Bromotoluene)'' | | [[Image:Benzyl-bromide-skeletal.svg|75px|Benzyl bromide]]<br />[[Benzyl bromide]]<br />''(1-Bromotoluene)'' | ||
|- | |- | ||
| Line 125: | Line 123: | ||
| [[Carboxylic acid|Carboxylate]] | | [[Carboxylic acid|Carboxylate]] | ||
| [[Carboxylate]] || RCOO<sup>−</sup> | | [[Carboxylate]] || RCOO<sup>−</sup> | ||
| − | | [[Image:Carboxylate-resonance-hybrid.png|75px|Carboxylate]]<br> | + | | [[Image:Carboxylate-resonance-hybrid.png|75px|Carboxylate]]<br/> |
[[Image:Carboxylate-canonical-forms.png|75px|Carboxylate]] | [[Image:Carboxylate-canonical-forms.png|75px|Carboxylate]] | ||
| carboxy- || -oate | | carboxy- || -oate | ||
| Line 134: | Line 132: | ||
| [[Image:Carboxylic-acid-skeletal.png|75px|Carboxylic acid]] | | [[Image:Carboxylic-acid-skeletal.png|75px|Carboxylic acid]] | ||
| carboxy- || -oic acid | | carboxy- || -oic acid | ||
| − | |||
|- | |- | ||
| rowspan="2" | [[Cyanate]]s | | rowspan="2" | [[Cyanate]]s | ||
| Line 154: | Line 151: | ||
| | | | ||
| alkyl alkyl disulfide | | alkyl alkyl disulfide | ||
| − | | [[Image:Cystamine.svg|130px|Cystamine]]<br>[[Cystamine]]<br>''(2,2'-Dithiobis(ethylamine))'' | + | | [[Image:Cystamine.svg|130px|Cystamine]]<br/>[[Cystamine]]<br/>''(2,2'-Dithiobis(ethylamine))'' |
|- | |- | ||
| − | | [[Ether]] || | + | | [[Ether]] || Ether |
| ROR' | | ROR' | ||
| [[Image:Ether-(general).png|75px|Ether]] | | [[Image:Ether-(general).png|75px|Ether]] | ||
| alkoxy- | | alkoxy- | ||
| alkyl alkyl '''ether''' | | alkyl alkyl '''ether''' | ||
| − | | [[Image:Diethyl-ether-2D-skeletal.png|75px|Diethyl ether]]<br>[[Diethyl ether]]<br>''(Ethoxyethane)'' | + | | [[Image:Diethyl-ether-2D-skeletal.png|75px|Diethyl ether]]<br/>[[Diethyl ether]]<br/>''(Ethoxyethane)'' |
|- | |- | ||
| − | | [[Ester]] || | + | | [[Ester]] || Ester |
| RCOOR' | | RCOOR' | ||
| [[Image:Ester-skeletal.png|Ester|75px]] | | [[Image:Ester-skeletal.png|Ester|75px]] | ||
| − | | | + | | || alkyl alkan'''oate''' |
| − | | [[Image:Ethyl butyrate.png|75px|Ethyl butyrate]]<br>[[Ethyl butyrate]]<br>''(Ethyl butanoate)'' | + | | [[Image:Ethyl butyrate.png|75px|Ethyl butyrate]]<br/>[[Ethyl butyrate]]<br/>''(Ethyl butanoate)'' |
|- | |- | ||
| [[Haloalkane]] || [[Halogen|Halo]] | | [[Haloalkane]] || [[Halogen|Halo]] | ||
| Line 173: | Line 170: | ||
| [[Image:Halide-group.png|75px|Halide group]] | | [[Image:Halide-group.png|75px|Halide group]] | ||
| halo- || alkyl '''halide''' | | halo- || alkyl '''halide''' | ||
| − | | [[Image:Chloroethane-skeletal.png|75px|Chloroethane]]<br>[[Chloroethane]]<br>''(Ethyl chloride)'' | + | | [[Image:Chloroethane-skeletal.png|75px|Chloroethane]]<br/>[[Chloroethane]]<br/>''(Ethyl chloride)'' |
|- | |- | ||
| [[Organic peroxide|Hydroperoxide]] | | [[Organic peroxide|Hydroperoxide]] | ||
| Line 208: | Line 205: | ||
| | | | ||
|- | |- | ||
| − | | [[Imide]] || | + | | [[Imide]] || Imide |
| RC(=O)NC(=O)R' | | RC(=O)NC(=O)R' | ||
| [[Image:Imide.png|75px|Imide]] | | [[Image:Imide.png|75px|Imide]] | ||
| Line 214: | Line 211: | ||
| -imide | | -imide | ||
|- | |- | ||
| − | | [[Isocyanide]] || | + | | [[Isocyanide]] || Isocyanide |
| RNC | | RNC | ||
| [[Image:Isocyanide-2D.png|75px|Isocyanide]] | | [[Image:Isocyanide-2D.png|75px|Isocyanide]] | ||
| Line 238: | Line 235: | ||
| [[Image:Ketone-skeletal.png|Ketone|75px]] | | [[Image:Ketone-skeletal.png|Ketone|75px]] | ||
| keto-, oxo- || -one | | keto-, oxo- || -one | ||
| − | | [[Image:Butanone-structure-skeletal.png|75px|Butanone]]<br />[[Methyl ethyl ketone]]<br>''(Butanone)'' | + | | [[Image:Butanone-structure-skeletal.png|75px|Butanone]]<br />[[Methyl ethyl ketone]]<br/>''(Butanone)'' |
|- | |- | ||
| − | | [[Nitrate]] || | + | | [[Nitrate]] || Nitrate |
| RONO<sub>2</sub> | | RONO<sub>2</sub> | ||
| [[Image:Nitrate-group-2D.png|75px|Nitrate]] | | [[Image:Nitrate-group-2D.png|75px|Nitrate]] | ||
| nitrooxy-, nitroxy- || | | nitrooxy-, nitroxy- || | ||
alkyl '''nitrate''' | alkyl '''nitrate''' | ||
| − | | [[Image:Amyl-nitrate-2D-skeletal.png|150px|Amyl nitrate]]<br/>[[Amyl nitrate]]<br>''(1-nitrooxypentane)'' | + | | [[Image:Amyl-nitrate-2D-skeletal.png|150px|Amyl nitrate]]<br/>[[Amyl nitrate]]<br/>''(1-nitrooxypentane)'' |
|- | |- | ||
| − | | [[Nitrile]] || | + | | [[Nitrile]] || Nitrile |
| RCN | | RCN | ||
| [[Image:Nitrile-(general)-skeletal.png|75px|Nitrile]] | | [[Image:Nitrile-(general)-skeletal.png|75px|Nitrile]] | ||
| cyano- || | | cyano- || | ||
| − | alkane'''nitrile'''<br> | + | alkane'''nitrile'''<br/> |
alkyl '''cyanide''' | alkyl '''cyanide''' | ||
| − | | [[Image:Benzonitrile structure.png|75px|Benzonitrile ]]<br />[[Benzonitrile]]<br>''(Phenyl cyanide)'' | + | | [[Image:Benzonitrile structure.png|75px|Benzonitrile ]]<br />[[Benzonitrile]]<br/>''(Phenyl cyanide)'' |
|- | |- | ||
| − | | [[Nitrite]] | | + | | [[Nitrite]] || Nitrosooxy |
| RONO | | RONO | ||
| [[Image:Nitrite-group-2D.png|75px|Nitrite]] | | [[Image:Nitrite-group-2D.png|75px|Nitrite]] | ||
| nitrosooxy- || | | nitrosooxy- || | ||
alkyl '''nitrite''' | alkyl '''nitrite''' | ||
| − | | [[Image:Amyl_nitrite.svg|150px|Amyl nitrite]]<br/>[[Amyl nitrite]]<br>''(3-methyl-1-nitrosooxybutane)'' | + | | [[Image:Amyl_nitrite.svg|150px|Amyl nitrite]]<br/>[[Amyl nitrite]]<br/>''(3-methyl-1-nitrosooxybutane)'' |
|- | |- | ||
| [[Nitro compound]] | | [[Nitro compound]] | ||
| Line 300: | Line 297: | ||
| HOPO(OR)<sub>2</sub> | | HOPO(OR)<sub>2</sub> | ||
| [[Image:Phosphodiester-group.png|75px|Phosphodiester]] | | [[Image:Phosphodiester-group.png|75px|Phosphodiester]] | ||
| − | | phosphoric acid di | + | | phosphoric acid di''(substituent)'' ester |
| − | | di | + | | di''(substituent)'' hydrogenphosphate |
| [[DNA]] | | [[DNA]] | ||
|- | |- | ||
| Line 321: | Line 318: | ||
| RC<sub>5</sub>H<sub>4</sub>N | | RC<sub>5</sub>H<sub>4</sub>N | ||
| | | | ||
| − | [[Image:4-pyridyl.png|75px|4-pyridyl group]]<br> | + | [[Image:4-pyridyl.png|75px|4-pyridyl group]]<br/> |
| − | [[Image:3-pyridyl.png|75px|3-pyridyl group]]<br> | + | [[Image:3-pyridyl.png|75px|3-pyridyl group]]<br/> |
| − | [[Image:2-pyridyl.png|75px|2-pyridyl group]]<br> | + | [[Image:2-pyridyl.png|75px|2-pyridyl group]]<br/> |
| | | | ||
| − | 4-pyridyl<br>(pyridin-4-yl)<br><br> | + | 4-pyridyl<br/>(pyridin-4-yl)<br/><br/> |
| − | 3-pyridyl<br>(pyridin-3-yl)<br><br> | + | 3-pyridyl<br/>(pyridin-3-yl)<br/><br/> |
| − | 2-pyridyl<br>(pyridin-2-yl) | + | 2-pyridyl<br/>(pyridin-2-yl) |
| -pyridine | | -pyridine | ||
| [[Image:Nicotine-2D-skeletal.png|75px|Nicotine]]<br />[[Nicotine]] | | [[Image:Nicotine-2D-skeletal.png|75px|Nicotine]]<br />[[Nicotine]] | ||
| Line 336: | Line 333: | ||
| [[Image:Sulfide-group-2D.png|75px|Sulfide group]] | | [[Image:Sulfide-group-2D.png|75px|Sulfide group]] | ||
| | | | ||
| − | | di | + | | di''(substituent)'' sulfide |
| [[Image:Dimethylsulfide.png|75px|Dimethyl sulfide]]<br />[[Dimethyl sulfide]] | | [[Image:Dimethylsulfide.png|75px|Dimethyl sulfide]]<br />[[Dimethyl sulfide]] | ||
|- | |- | ||
| Line 344: | Line 341: | ||
| [[Image:Sulfone.png|75px|Sulfonyl group]] | | [[Image:Sulfone.png|75px|Sulfonyl group]] | ||
| sulfonyl- | | sulfonyl- | ||
| − | | di | + | | di''(substituent)'' sulfone |
| − | | [[Image:Dimethylsulfone_Structure.png|75px|Dimethyl sulfone]]<br />Dimethyl sulfone<br>''([[Methylsulfonylmethane]])'' | + | | [[Image:Dimethylsulfone_Structure.png|75px|Dimethyl sulfone]]<br />Dimethyl sulfone<br/>''([[Methylsulfonylmethane]])'' |
|- | |- | ||
| [[Sulfonic acid]] || Sulfo | | [[Sulfonic acid]] || Sulfo | ||
| Line 359: | Line 356: | ||
| [[Image:Sulfoxide.png|75px|Sulfinyl group]] | | [[Image:Sulfoxide.png|75px|Sulfinyl group]] | ||
| sulfinyl- | | sulfinyl- | ||
| − | | di | + | | di''(substituent)'' '''sulfoxide''' |
| [[Image:Diphenyl-sulfoxide.png|75px|Diphenyl sulfoxide]]<br />Diphenyl sulfoxide | | [[Image:Diphenyl-sulfoxide.png|75px|Diphenyl sulfoxide]]<br />Diphenyl sulfoxide | ||
|- | |- | ||
| Line 372: | Line 369: | ||
==See also== | ==See also== | ||
| − | * [[ | + | |
| + | * [[Chemical classification]] | ||
| + | * [[Organic chemistry]] | ||
| + | |||
| + | == References == | ||
| + | |||
| + | * McMurry, John. ''Organic Chemistry''. 6th ed. Belmont, CA: Brooks/Cole, 2004. ISBN 0534420052 | ||
| + | * Morrison, Robert T., and Robert N. Boyd. ''Organic Chemistry''. 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0-13-643669-2 | ||
| + | * Solomons, T.W. Graham, and Fryhle, Craig B. ''Organic Chemistry''. 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998 | ||
==External links== | ==External links== | ||
| − | + | All links retrieved May 16, 2017. | |
| − | * [http:// | + | * [http://goldbook.iupac.org/F02555.html IUPAC Compendium of Chemical Terminology - the Gold Book] ''International Union of Pure and Applied Chemistry''. |
| + | {{Functional Groups}} | ||
| − | |||
{{Organic chemistry}} | {{Organic chemistry}} | ||
[[Category:Physical sciences]] | [[Category:Physical sciences]] | ||
[[Category:Chemistry]] | [[Category:Chemistry]] | ||
| − | [[Category:Organic | + | [[Category:Organic chemistry]] |
{{credit|133462962}} | {{credit|133462962}} | ||
Revision as of 10:43, 21 June 2021
In organic chemistry, functional groups (or moieties) are specific groups of atoms within molecules, that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction(s) regardless of the size of the molecule it is a part of.
Combining the names of functional groups with the names of the parent alkanes generates a powerful systematic nomenclature for naming organic compounds.
The non-hydrogen atoms of functional groups are always associated with each other and with the rest of the molecule by covalent bonds. When the group of atoms is associated with the rest of the molecule primarily by ionic forces, the group is referred to more properly as a polyatomic ion or complex ion—all of these are called radicals, by a meaning of the term radical that predates the free radical.
The first carbon atom after the carbon that attaches to the functional group is called the alpha carbon.
Functional groups are attached to the carbon backbone of organic molecules. They determine the characteristics and chemical reactivity of molecules. Functional groups are far less stable than the carbon backbone and are likely to participate in chemical reactions.
Table of common functional groups
The following is a list of common functional groups. In the formulas, the symbols R and R' usually denotes an attached hydrogen, or a hydrocarbon side chain of any length, but may sometimes refer to any group of atoms. Below is an image of multiple functional groups found in organic chemistry.
(For convenience, see basic functional groups covered in General Biology)
| Chemical class | Group | Formula | Structural Formula | Prefix | Suffix | Example |
|---|---|---|---|---|---|---|
| Acyl halide | Haloformyl | RCOX | 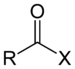
|
haloformyl- | -oyl halide | 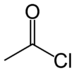 Acetyl chloride (Ethanoyl chloride) |
| Alcohol | Hydroxyl | ROH | 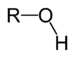
|
hydroxy- | -ol | 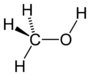 Methanol |
| Aldehyde | Aldehyde | RCHO | 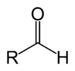
|
aldo- | -al | 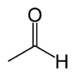 Acetaldehyde (Ethanal) |
| Alkane | Alkyl | RH | alkyl- | -ane | 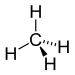 Methane | |
| Alkene | Alkenyl | R2C=CR2 | Alkene | alkenyl- | -ene |  Ethylene (Ethene) |
| Alkyne | Alkynyl | RC≡CR' | alkynyl- | -yne | ||
| Amide | Carboxamide | RCONR2 | 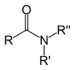
|
carboxamido- | -amide | |
| Amines | Primary amine | RNH2 | amino- | -amine | 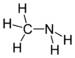 Methylamine (Methanamine) | |
| Secondary amine | R2NH | 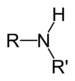
|
amino- | -amine | Dimethylamine | |
| Tertiary amine | R3N | 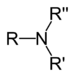
|
amino- | -amine | 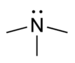 Trimethylamine | |
| 4° ammonium ion | R4N+ | 
|
ammonio- | -ammonium | 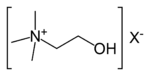 Choline | |
| Azide | Azide | RN3 | azido- | alkyl azide | 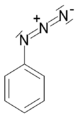 Phenyl azide (Azidobenzene) | |
| Azo compound | Azo (Diimide) |
RN2R' | 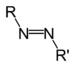
|
azo- | -diazene | 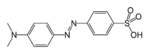 Methyl orange (p-dimethylamino-azobenzenesulfonic acid) |
| Toluene derivative | Benzyl | RCH2C6H5 RBn |
benzyl- | 1-(substituent)toluene | Benzyl bromide (1-Bromotoluene) | |
| Carbonate | Carbonate ester | ROCOOR | alkyl carbonate | |||
| Carboxylate | Carboxylate | RCOO− | 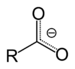 |
carboxy- | -oate | Sodium acetate (Sodium ethanoate) |
| Carboxylic acid | Carboxyl | RCOOH | 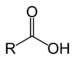
|
carboxy- | -oic acid | |
| Cyanates | Cyanate | ROCN | cyanato- | alkyl cyanate | ||
| Thiocyanate | RSCN | thiocyanato- | alkyl thiocyanate | |||
| Disulfide | Disulfide | RSSR' | alkyl alkyl disulfide | Cystamine (2,2'-Dithiobis(ethylamine)) | ||
| Ether | Ether | ROR' | alkoxy- | alkyl alkyl ether | Diethyl ether (Ethoxyethane) | |
| Ester | Ester | RCOOR' | 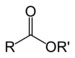
|
alkyl alkanoate | Ethyl butyrate (Ethyl butanoate) | |
| Haloalkane | Halo | RX | halo- | alkyl halide | Chloroethane (Ethyl chloride) | |
| Hydroperoxide | Hydroperoxy | ROOH | hydroperoxy- | alkyl hydroperoxide | Methyl ethyl ketone peroxide | |
| Imine | Primary ketimine | RC(=NH)R' | 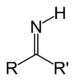
|
imino- | -imine | |
| Secondary ketimine | RC(=NR)R' | 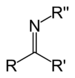
|
imino- | -imine | ||
| Primary aldimine | RC(=NH)H | 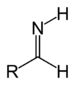
|
imino- | -imine | ||
| Secondary aldimine | RC(=NR')H | 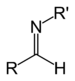
|
imino- | -imine | ||
| Imide | Imide | RC(=O)NC(=O)R' | 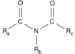
|
imido- | -imide | |
| Isocyanide | Isocyanide | RNC | isocyano- | alkyl isocyanide | ||
| Isocyanates | Isocyanate | RNCO | isocyanato- | alkyl isocyanate | Methyl isocyanate | |
| Isothiocyanate | RNCS | isothiocyanato- | alkyl isothiocyanate | Allyl isothiocyanate | ||
| Ketone | Carbonyl | RCOR' | 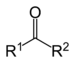
|
keto-, oxo- | -one | 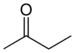 Methyl ethyl ketone (Butanone) |
| Nitrate | Nitrate | RONO2 | nitrooxy-, nitroxy- |
alkyl nitrate |
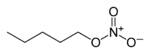 Amyl nitrate (1-nitrooxypentane) | |
| Nitrile | Nitrile | RCN | cyano- |
alkanenitrile |
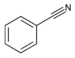 Benzonitrile (Phenyl cyanide) | |
| Nitrite | Nitrosooxy | RONO | nitrosooxy- |
alkyl nitrite |
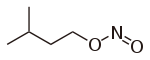 Amyl nitrite (3-methyl-1-nitrosooxybutane) | |
| Nitro compound | Nitro | RNO2 | 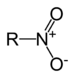
|
nitro- | 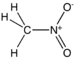 Nitromethane | |
| Nitroso compound | Nitroso | RNO | nitroso- | Nitrosobenzene | ||
| Peroxide | Peroxy | ROOR | 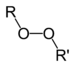
|
peroxy- | alkyl peroxide | Di-tert-butyl peroxide |
| Benzene derivative | Phenyl | RC6H5 | phenyl- | -benzene | 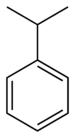 Cumene (2-phenylpropane) | |
| Phosphine | Phosphino | R3P | 
|
phosphino- | -phosphane | Methylpropylphosphane |
| Phosphodiester | Phosphate | HOPO(OR)2 | 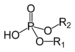
|
phosphoric acid di(substituent) ester | di(substituent) hydrogenphosphate | DNA |
| Phosphonic acid | Phosphono | RP(=O)(OH)2 | 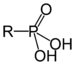
|
phosphono- | substituent phosphonic acid | Benzylphosphonic acid |
| Phosphate | Phosphate | ROP(=O)(OH)2 | 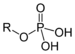
|
phospho- | Glyceraldehyde 3-phosphate | |
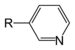
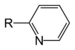
| Pyridine derivative | Pyridyl | RC5H4N |
4-pyridyl |
-pyridine |  Nicotine | |
| Sulfide | RSR' | di(substituent) sulfide | Dimethyl sulfide | |||
| Sulfone | Sulfonyl | RSO2R' | 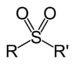
|
sulfonyl- | di(substituent) sulfone | 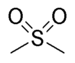 Dimethyl sulfone (Methylsulfonylmethane) |
| Sulfonic acid | Sulfo | RSO3H | 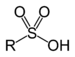
|
sulfo- | substituent sulfonic acid | 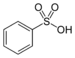 Benzenesulfonic acid |
| Sulfoxide | Sulfinyl | RSOR' | 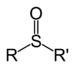
|
sulfinyl- | di(substituent) sulfoxide | 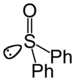 Diphenyl sulfoxide |
| Thiol | Sulfhydryl | RSH | 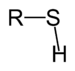
|
mercapto-, sulfanyl- | -thiol | Ethanethiol (Ethyl mercaptan) |
See also
- Chemical classification
- Organic chemistry
ReferencesISBN links support NWE through referral fees
- McMurry, John. Organic Chemistry. 6th ed. Belmont, CA: Brooks/Cole, 2004. ISBN 0534420052
- Morrison, Robert T., and Robert N. Boyd. Organic Chemistry. 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0-13-643669-2
- Solomons, T.W. Graham, and Fryhle, Craig B. Organic Chemistry. 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998
External links
All links retrieved May 16, 2017.
- IUPAC Compendium of Chemical Terminology - the Gold Book International Union of Pure and Applied Chemistry.
| Functional groups |
|---|
| Chemical class: Alcohol • Aldehyde • Alkane • Alkene • Alkyne • Amide • Amine • Azo compound • Benzene derivative • Carboxylic acid • Cyanate • Ester • Ether • Haloalkane • Imine • Isocyanide • Isocyanate • Ketone • Nitrile • Nitro compound • Nitroso compound • Peroxide • Phosphoric acid • Pyridine derivative • Sulfone • Sulfonic acid • Sulfoxide • Thioether • Thiol • Toluene derivative |
| Topics in organic chemistry |
|---|
|
Aromaticity | Covalent bonding | Functional groups | Nomenclature | Organic compounds | Organic reactions | Organic synthesis | Publications | Spectroscopy | Stereochemistry |
| List of organic compounds |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.