Difference between revisions of "Acetylcholine" - New World Encyclopedia
(New page: {{Claimed}}) |
|||
| Line 1: | Line 1: | ||
{{Claimed}} | {{Claimed}} | ||
| + | {{drugbox | ||
| + | | IUPAC_name = 2-acetoxy-N,N,N-trimethylethanaminium | ||
| + | | image = ACh.png | ||
| + | | width = 200 | ||
| + | | image2 = ACh-stick.png | ||
| + | | width2 = 200 | ||
| + | | CAS_number = 51-84-3 | ||
| + | | ATC_prefix = S01 | ||
| + | | ATC_suffix = EB09 | ||
| + | | ATC_supplemental = | ||
| + | | PubChem = 187 | ||
| + | | DrugBank = EXPT00412 | ||
| + | | chemical_formula = | ||
| + | | C=7 | H=16 | N=1 | O=2 | ||
| + | | molecular_weight = 146.21 g/mol | ||
| + | | smiles = CC(OCC[N<sup>+</sup>](C)(C)C)=O | ||
| + | | bioavailability = | ||
| + | | protein_bound = | ||
| + | | metabolism = | ||
| + | | elimination_half-life = approximately 2 minutes | ||
| + | | pregnancy_category = | ||
| + | | legal_status = | ||
| + | | routes_of_administration = | ||
| + | }} | ||
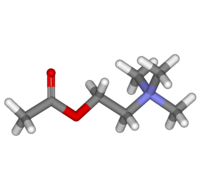
| + | The [[chemical compound]] '''acetylcholine''', often abbreviated as '''ACh''', was the first [[neurotransmitter]] to be identified. It is a chemical transmitter in both the [[peripheral nervous system]] (PNS) and [[central nervous system]] (CNS) in many organisms including [[homo sapiens|humans]]. Acetylcholine is the neurotransmitter in all [[autonomic ganglia]]. In layman's terms, it is a chemical that allows neurons to communicate with each other within humans and other organisms. | ||
| + | ==Chemistry== | ||
| + | Acetylcholine is an [[ester]] of [[acetic acid]] and [[choline]] with [[chemical formula]] [[acetyl|CH<sub><small>3</small></sub>CO]][[oxygen|O]][[methylene|CH<sub><small>2</small></sub>]][[methylene|CH<sub><small>2</small></sub>]][[Nitrogen|N]]<sup><small>+</small></sup>([[methyl|CH<sub><small>3</small></sub>]])<sub><small>3</small></sub>. This structure is reflected in the systematic name, ''2-acetoxy-N,N,N-trimethylethanaminium''. | ||
| + | |||
| + | Acetylcholine (ACh) was first identified in 1914 by [[Henry Hallett Dale]] for its actions on heart tissue. It was confirmed as a neurotransmitter by [[Otto Loewi]] who initially gave it the name [[vagusstoff]] because it was released from the [[vagus nerve]]. Both received the 1936 [[Nobel Prize in Physiology or Medicine]] for their work. | ||
| + | |||
| + | Later work showed that when acetylcholine binds to [[acetylcholine receptor]]s on [[skeletal muscle]] fibers, it opens ligand gated sodium channels in the membrane. Sodium ions then enter the muscle cell, stimulating muscle contraction. Acetylcholine, while inducing contraction of skeletal muscles, instead induces decreased contraction in [[cardiac muscle]] fibers. This distinction is attributed to differences in receptor structure between skeletal and cardiac fibers. Acetylcholine is also used in the [[brain]], where it tends to cause excitatory actions. The [[gland]]s that receive impulses from the parasympathetic part of the [[autonomic nervous system]] are also stimulated in the same way. | ||
| + | |||
| + | ==Synthesis and Degradation== | ||
| + | Acetylcholine is synthesized in certain [[neuron]]s by the [[enzyme]] [[choline acetyltransferase]] from the compounds [[choline]] and [[acetyl-CoA]]. Organic mercurial compounds have a high affinity for [[thiol|sulfhydryl groups]], which causes dysfunction of the enzyme choline acetyl transferase. This inhibition may lead to acetylcholine deficiency, and can have consequences on motor function. | ||
| + | |||
| + | Normally, the enzyme [[acetylcholinesterase]] converts acetylcholine into the inactive [[metabolites]] [[choline]] and [[acetate]]. This enzyme is abundant in the synaptic cleft, and its role in rapidly clearing free acetylcholine from the synapse is essential for proper muscle function. The devastating effects of [[nerve agent|organophosphate-containing nerve agents]] (e.g. [[Sarin gas]]) are due to their irreversible inactivation of this enzyme. The resulting accumulation of acetylcholine causes continuous stimulation of the muscles, glands and central nervous system; victims commonly die of suffocation as they cannot contract their [[diaphragm]]. Other organophosphates and some [[carbamates]] are effective insecticides because they inhibit acetylcholinasterase in insects. On the other hand, since a shortage of acetylcholine in the [[brain]] has been associated with [[Alzheimer's disease]], some drugs that inhibit acetylcholinesterase are used in the treatment of that disease. A recent study [http://www.msnbc.msn.com/id/15145917/] has shown that [[THC]] is one such drug, effective at reducing the formation of characteristic [[neurofibrillary tangle]]s and [[amyloid beta]] plaques. | ||
| + | |||
| + | ==Release sites== | ||
| + | *Acetylcholine is released in the [[autonomic nervous system]]: | ||
| + | **pre- and post-ganglionic [[parasympathetic nervous system|parasympathetic]] neurons | ||
| + | **preganglionic [[sympathetic nervous system|sympathetic]] neurons (and also postganglionic sudomotor neurons, i.e., the ones that control [[sweating]]) | ||
| + | |||
| + | [[Botulism toxin|Botulin]] acts by suppressing the release of acetylcholine; where the venom from a [[black widow spider]] has the reverse effect. | ||
| + | |||
| + | *all preganglionic autonomic fibers including: | ||
| + | **all preganglionic sympathetic fibers | ||
| + | **all preganglionic parasympathetic fibers | ||
| + | **preganglionic sympathetic fibers to suprarenal medulla, the modified sympathetic ganglion. On stimulation by acetylcholine, it releases [[adrenaline]] and [[noradrenaline]]. | ||
| + | *all postganglionic parasympathetic fibers | ||
| + | *some postganglionic sympathetic fibers | ||
| + | **secretory fibers to sweat glands | ||
| + | **vasodilator fibers to blood vessels of skeletal muscles | ||
| + | |||
| + | ==Pharmacology== | ||
| + | There are two main classes of acetylcholine receptor (AChR), [[nicotinic acetylcholine receptor]]s (nAChR) and [[muscarinic acetylcholine receptor]]s (mAChR). They are named for the [[ligands]] used to discover the receptors. | ||
| + | |||
| + | Nicotinic AChRs are [[Ligand-gated ion channel|ionotropic receptors]] permeable to [[sodium]], [[potassium]], and [[chloride]] ions. They are stimulated by [[nicotine]] and acetylcholine and blocked by [[curare]]. Most peripheral AChRs are nicotinic, such as those on the heart and blood vessels or at the [[neuromuscular junction]]. They are also found in wide distribution through the brain, but in relatively low numbers. | ||
| + | |||
| + | Muscarinic receptors are [[metabotropic]] and affect neurons over a longer time frame. They are stimulated by [[muscarine]] and acetylcholine, and blocked by [[atropine]]. Muscarinic receptors are found in both the central nervous system and the peripheral nervous system, in heart, lungs, upper GI tract and sweat glands. Extracts from the plant included this compound, and its action on muscarinic AChRs that increased pupil size was used for attractiveness in many European cultures in the past. Now, ACh is sometimes used during [[cataract]] surgery to produce rapid constriction of the pupil. It must be administered intraocularly because [[cornea|corneal]] [[cholinesterase]] metabolizes topically administered ACh before it can diffuse into the eye. It is sold by the trade name Miochol-E (CIBA Vision). Similar drugs are used to induce [[mydriasis]] (dilation of the pupil) in [[cardiopulmonary resuscitation]] and many other situations. | ||
| + | |||
| + | The disease [[myasthenia gravis]], characterized by muscle weakness and fatigue, occurs when the body inappropriately produces [[antibody|antibodies]] against acetylcholine receptors, and thus inhibits proper acetylcholine signal transmission. Over time the motor end plate is destroyed. Drugs that competitively inhibit acetylcholinesterase (e.g., neostigmine or physostigmine) are effective in treating this disorder. They allow endogenously released acetylcholine more time to interact with its respective receptor before being inactivated by acetylcholinesterase in the gap junction. | ||
| + | |||
| + | Blocking, hindering or mimicking the action of acetylcholine has many uses in medicine. [[Cholinesterase inhibitor]]s, an example of [[enzyme inhibitors]], increase the action of acetylcholine by delaying its degradation; some have been used as [[nerve agent]]s ([[Sarin]] and [[VX (nerve agent)|VX]] nerve gas) or [[pesticide]]s ([[organophosphates]] and the [[carbamates]]). Clinically they are used to reverse the action of [[muscle relaxant]]s, to treat [[myasthenia gravis]] and in [[Alzheimer's disease]] ([[rivastigmine]], which increases cholinergic activity in the brain). | ||
| + | |||
| + | === ACh Receptor Agonists === | ||
| + | ====Direct Acting==== | ||
| + | * Acetylcholine | ||
| + | * [[Bethanechol]] | ||
| + | * [[Carbachol]] | ||
| + | * [[Cevimeline]] | ||
| + | * [[Pilocarpine]] | ||
| + | * [[Suberylcholine]] | ||
| + | |||
| + | ====Indirect Acting (reversible)==== | ||
| + | Reversibly inhibit the enzyme [[acetylcholinesterase]] (which breaks down acetylcholine), thereby increasing acetylcholine levels. | ||
| + | * [[Ambenomium]] | ||
| + | * [[Donepezil]] | ||
| + | * [[Edrophonium]] | ||
| + | * [[Galantamine]] | ||
| + | * [[Neostigmine]] | ||
| + | * [[Physostigmine]] | ||
| + | * [[Pyridostigmine]] | ||
| + | * [[Rivastigmine]] | ||
| + | * [[Tacrine]] | ||
| + | * [[Carbamate]] [[Insecticides]] ([[Aldicarb]]) | ||
| + | |||
| + | ====Indirect Acting (irreversible)==== | ||
| + | Semi-permanently inhibit the enzyme acetylcholinesterase. | ||
| + | * [[Echothiophate]] | ||
| + | * [[Isoflurophate]] | ||
| + | * [[Organophosphate]] [[Insecticides]] ([[Malathion]], [[Parathion]], [[Azinphos Methyl]], [[Chlorpyrifos]], among others) | ||
| + | |||
| + | ====Reactivation of Acetylcholine Esterase==== | ||
| + | * [[Pralidoxime]] | ||
| + | |||
| + | === ACh Receptor Antagonists === | ||
| + | ====Antimuscarinic Agents==== | ||
| + | * [[Atropine]] | ||
| + | * [[Ipratropium]] | ||
| + | * [[Scopolamine]] | ||
| + | * [[Tiotropium]] | ||
| + | |||
| + | ====Ganglionic Blockers==== | ||
| + | * [[Mecamylamine]] | ||
| + | * [[Hexamethonium]] | ||
| + | * [[Nicotine]] (in high doses) | ||
| + | * [[Trimethaphan]] | ||
| + | |||
| + | ====Neuromuscular Blockers==== | ||
| + | * [[Atracurium]] | ||
| + | * [[Cisatracurium]] | ||
| + | * [[Doxacurium]] | ||
| + | * [[Metocurine]] | ||
| + | * [[Mivacurium]] | ||
| + | * [[Pancuronium]] | ||
| + | * [[Rocuronium]] | ||
| + | * [[Succinylcholine]] | ||
| + | * [[Tubovurarine]] | ||
| + | * [[Vecuronium]] | ||
| + | |||
| + | ====Others? / Uncategorized / Unknown==== | ||
| + | * [[surugatoxin]] | ||
| + | * Organophosphates block the breakdown of acetylcholine. Tetrachlorvinphos and other organophosphates operate by blocking acetylcholinesterase, which is an enzyme that breaks down acetylcholine. | ||
| + | |||
| + | == Neuromodulatory Effects == | ||
| + | In the central nervous system, ACh has a variety of effects as a [[neuromodulator]]. | ||
| + | |||
| + | Given its prominent role in learning, ACh is naturally involved with [[synaptic plasticity]]. It has been shown to enhance the amplitude of synaptic potentials following [[long-term potentiation]] in many regions, including the [[dentate gyrus]], [[CA1]], [[piriform cortex]], and [[neocortex]]. This effect most likely occurs either through enhancing currents through [[NMDA]] receptors or indirectly by suppressing [[neural adaptation|adaptation]]. The suppression of adaptation has been shown in brain slices of regions CA1, [[cingulate cortex]], and piriform cortex as well as [[in vivo]] in cat [[somatosensory cortex|somatosensory]] and [[motor cortex]] by decreasing the conductance of voltage-dependent [[M current]]s and [[calcium|Ca<sup>2+</sup>]]-dependent [[potassium|K<sup>+</sup>]] currents. | ||
| + | |||
| + | Acetylcholine also has other effects on excitability of neurons. Its presence causes a slow [[depolarization]] by blocking a tonically active K<sup>+</sup> current, which increases neuronal excitability. Paradoxically, it increases spiking activity in [[inhibitory interneuron]]s while decreasing strength of synaptic transmission from those cells. This decrease in synaptic transmission also occurs selectively at some excitatory cells: for instance, it has an effect on intrinsic and associational fibers in layer Ib of piriform cortex, but has no effect on afferent fibers in layer Ia. Similar laminar selectivity has been shown in dentate gyrus and region CA1 of the hippocampus. One theory to explain this paradox interprets Acetylcholine neuromodulation in the neocortex as modulating the estimate of expected uncertainty, acting counter to [[Norepinephrine]] (NE) signals for unexpected uncertainty. Both would then decrease synaptic transition strength, but ACh would then be needed to counter the effects of NE in learning a signal understood to be noisy. | ||
| + | |||
| + | ==Sources== | ||
| + | *Brenner, G. M. and Stevens, C. W. (2006). ''Pharmacology, 2nd Edition.'' Philadelphia, PA: W.B. Saunders Company (Elsevier). ISBN 1-4160-2984-2 | ||
| + | *Canadian Pharmacists Association (2000). Compendium of Pharmaceuticals and Specialties (25th ed.). Toronto, ON: Webcom. ISBN 0-919115-76-4 | ||
| + | *Carlson, NR (2001). Physiology of Behavior-7th ed. Needham Heights, MA: Allyn and Bacon. ISBN 0-205-30840-6 | ||
| + | *Gershon, Michael D. (1998). The Second Brain. New York, NY: HarperCollins. ISBN 0-06-018252-0 | ||
| + | *Hasselmo, ME (1995). Neuromodulation and cortical function: Modeling the physiological basis of behavior. Behav. Brain Res. 67: 1-27 [http://people.bu.edu/hasselmo/HasselmoBBR1995.pdf] | ||
| + | *Yu, AJ & [[Peter Dayan|Dayan, P]] (2005). ''Uncertainty, neuromodulation, and attention.'' Neuron 46 681-692. [http://www.gatsby.ucl.ac.uk/~dayan/papers/yud2005.pdf] | ||
| + | |||
| + | ==External links== | ||
| + | * [http://www.neuro.wustl.edu/neuromuscular/mother/acetylcholine.htm Washington University (St. Louis) writeup] | ||
| + | |||
| + | {{ChemicalSources}} | ||
| + | |||
| + | [[Category:Choline esters]] | ||
| + | [[Category:Acetates]] | ||
| + | [[Category:Neurotransmitters]] | ||
| + | [[Category:Quaternary ammonium compounds]] | ||
| + | {{credit|135117125}} | ||
| + | [[Category:Life sciences]] | ||
Revision as of 16:05, 15 June 2007

| |
| |
Acetylcholine
| |
| Systematic name | |
| IUPAC name 2-acetoxy-N,N,N-trimethylethanaminium | |
| Identifiers | |
| CAS number | 51-84-3 |
| ATC code | S01EB09 |
| PubChem | 187 |
| DrugBank | EXPT00412 |
| Chemical data | |
| Formula | C7H16NO2 |
| Mol. weight | 146.21 g/mol |
| SMILES | CC(OCC[N+](C)(C)C)=O |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | approximately 2 minutes |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. | ? |
| Legal status | ? |
| Routes | ? |
The chemical compound acetylcholine, often abbreviated as ACh, was the first neurotransmitter to be identified. It is a chemical transmitter in both the peripheral nervous system (PNS) and central nervous system (CNS) in many organisms including humans. Acetylcholine is the neurotransmitter in all autonomic ganglia. In layman's terms, it is a chemical that allows neurons to communicate with each other within humans and other organisms.
Chemistry
Acetylcholine is an ester of acetic acid and choline with chemical formula CH3COOCH2CH2N+(CH3)3. This structure is reflected in the systematic name, 2-acetoxy-N,N,N-trimethylethanaminium.
Acetylcholine (ACh) was first identified in 1914 by Henry Hallett Dale for its actions on heart tissue. It was confirmed as a neurotransmitter by Otto Loewi who initially gave it the name vagusstoff because it was released from the vagus nerve. Both received the 1936 Nobel Prize in Physiology or Medicine for their work.
Later work showed that when acetylcholine binds to acetylcholine receptors on skeletal muscle fibers, it opens ligand gated sodium channels in the membrane. Sodium ions then enter the muscle cell, stimulating muscle contraction. Acetylcholine, while inducing contraction of skeletal muscles, instead induces decreased contraction in cardiac muscle fibers. This distinction is attributed to differences in receptor structure between skeletal and cardiac fibers. Acetylcholine is also used in the brain, where it tends to cause excitatory actions. The glands that receive impulses from the parasympathetic part of the autonomic nervous system are also stimulated in the same way.
Synthesis and Degradation
Acetylcholine is synthesized in certain neurons by the enzyme choline acetyltransferase from the compounds choline and acetyl-CoA. Organic mercurial compounds have a high affinity for sulfhydryl groups, which causes dysfunction of the enzyme choline acetyl transferase. This inhibition may lead to acetylcholine deficiency, and can have consequences on motor function.
Normally, the enzyme acetylcholinesterase converts acetylcholine into the inactive metabolites choline and acetate. This enzyme is abundant in the synaptic cleft, and its role in rapidly clearing free acetylcholine from the synapse is essential for proper muscle function. The devastating effects of organophosphate-containing nerve agents (e.g. Sarin gas) are due to their irreversible inactivation of this enzyme. The resulting accumulation of acetylcholine causes continuous stimulation of the muscles, glands and central nervous system; victims commonly die of suffocation as they cannot contract their diaphragm. Other organophosphates and some carbamates are effective insecticides because they inhibit acetylcholinasterase in insects. On the other hand, since a shortage of acetylcholine in the brain has been associated with Alzheimer's disease, some drugs that inhibit acetylcholinesterase are used in the treatment of that disease. A recent study [1] has shown that THC is one such drug, effective at reducing the formation of characteristic neurofibrillary tangles and amyloid beta plaques.
Release sites
- Acetylcholine is released in the autonomic nervous system:
- pre- and post-ganglionic parasympathetic neurons
- preganglionic sympathetic neurons (and also postganglionic sudomotor neurons, i.e., the ones that control sweating)
Botulin acts by suppressing the release of acetylcholine; where the venom from a black widow spider has the reverse effect.
- all preganglionic autonomic fibers including:
- all preganglionic sympathetic fibers
- all preganglionic parasympathetic fibers
- preganglionic sympathetic fibers to suprarenal medulla, the modified sympathetic ganglion. On stimulation by acetylcholine, it releases adrenaline and noradrenaline.
- all postganglionic parasympathetic fibers
- some postganglionic sympathetic fibers
- secretory fibers to sweat glands
- vasodilator fibers to blood vessels of skeletal muscles
Pharmacology
There are two main classes of acetylcholine receptor (AChR), nicotinic acetylcholine receptors (nAChR) and muscarinic acetylcholine receptors (mAChR). They are named for the ligands used to discover the receptors.
Nicotinic AChRs are ionotropic receptors permeable to sodium, potassium, and chloride ions. They are stimulated by nicotine and acetylcholine and blocked by curare. Most peripheral AChRs are nicotinic, such as those on the heart and blood vessels or at the neuromuscular junction. They are also found in wide distribution through the brain, but in relatively low numbers.
Muscarinic receptors are metabotropic and affect neurons over a longer time frame. They are stimulated by muscarine and acetylcholine, and blocked by atropine. Muscarinic receptors are found in both the central nervous system and the peripheral nervous system, in heart, lungs, upper GI tract and sweat glands. Extracts from the plant included this compound, and its action on muscarinic AChRs that increased pupil size was used for attractiveness in many European cultures in the past. Now, ACh is sometimes used during cataract surgery to produce rapid constriction of the pupil. It must be administered intraocularly because corneal cholinesterase metabolizes topically administered ACh before it can diffuse into the eye. It is sold by the trade name Miochol-E (CIBA Vision). Similar drugs are used to induce mydriasis (dilation of the pupil) in cardiopulmonary resuscitation and many other situations.
The disease myasthenia gravis, characterized by muscle weakness and fatigue, occurs when the body inappropriately produces antibodies against acetylcholine receptors, and thus inhibits proper acetylcholine signal transmission. Over time the motor end plate is destroyed. Drugs that competitively inhibit acetylcholinesterase (e.g., neostigmine or physostigmine) are effective in treating this disorder. They allow endogenously released acetylcholine more time to interact with its respective receptor before being inactivated by acetylcholinesterase in the gap junction.
Blocking, hindering or mimicking the action of acetylcholine has many uses in medicine. Cholinesterase inhibitors, an example of enzyme inhibitors, increase the action of acetylcholine by delaying its degradation; some have been used as nerve agents (Sarin and VX nerve gas) or pesticides (organophosphates and the carbamates). Clinically they are used to reverse the action of muscle relaxants, to treat myasthenia gravis and in Alzheimer's disease (rivastigmine, which increases cholinergic activity in the brain).
ACh Receptor Agonists
Direct Acting
- Acetylcholine
- Bethanechol
- Carbachol
- Cevimeline
- Pilocarpine
- Suberylcholine
Indirect Acting (reversible)
Reversibly inhibit the enzyme acetylcholinesterase (which breaks down acetylcholine), thereby increasing acetylcholine levels.
- Ambenomium
- Donepezil
- Edrophonium
- Galantamine
- Neostigmine
- Physostigmine
- Pyridostigmine
- Rivastigmine
- Tacrine
- Carbamate Insecticides (Aldicarb)
Indirect Acting (irreversible)
Semi-permanently inhibit the enzyme acetylcholinesterase.
- Echothiophate
- Isoflurophate
- Organophosphate Insecticides (Malathion, Parathion, Azinphos Methyl, Chlorpyrifos, among others)
Reactivation of Acetylcholine Esterase
- Pralidoxime
ACh Receptor Antagonists
Antimuscarinic Agents
- Atropine
- Ipratropium
- Scopolamine
- Tiotropium
Ganglionic Blockers
- Mecamylamine
- Hexamethonium
- Nicotine (in high doses)
- Trimethaphan
Neuromuscular Blockers
- Atracurium
- Cisatracurium
- Doxacurium
- Metocurine
- Mivacurium
- Pancuronium
- Rocuronium
- Succinylcholine
- Tubovurarine
- Vecuronium
Others? / Uncategorized / Unknown
- surugatoxin
- Organophosphates block the breakdown of acetylcholine. Tetrachlorvinphos and other organophosphates operate by blocking acetylcholinesterase, which is an enzyme that breaks down acetylcholine.
Neuromodulatory Effects
In the central nervous system, ACh has a variety of effects as a neuromodulator.
Given its prominent role in learning, ACh is naturally involved with synaptic plasticity. It has been shown to enhance the amplitude of synaptic potentials following long-term potentiation in many regions, including the dentate gyrus, CA1, piriform cortex, and neocortex. This effect most likely occurs either through enhancing currents through NMDA receptors or indirectly by suppressing adaptation. The suppression of adaptation has been shown in brain slices of regions CA1, cingulate cortex, and piriform cortex as well as in vivo in cat somatosensory and motor cortex by decreasing the conductance of voltage-dependent M currents and Ca2+-dependent K+ currents.
Acetylcholine also has other effects on excitability of neurons. Its presence causes a slow depolarization by blocking a tonically active K+ current, which increases neuronal excitability. Paradoxically, it increases spiking activity in inhibitory interneurons while decreasing strength of synaptic transmission from those cells. This decrease in synaptic transmission also occurs selectively at some excitatory cells: for instance, it has an effect on intrinsic and associational fibers in layer Ib of piriform cortex, but has no effect on afferent fibers in layer Ia. Similar laminar selectivity has been shown in dentate gyrus and region CA1 of the hippocampus. One theory to explain this paradox interprets Acetylcholine neuromodulation in the neocortex as modulating the estimate of expected uncertainty, acting counter to Norepinephrine (NE) signals for unexpected uncertainty. Both would then decrease synaptic transition strength, but ACh would then be needed to counter the effects of NE in learning a signal understood to be noisy.
Sources
- Brenner, G. M. and Stevens, C. W. (2006). Pharmacology, 2nd Edition. Philadelphia, PA: W.B. Saunders Company (Elsevier). ISBN 1-4160-2984-2
- Canadian Pharmacists Association (2000). Compendium of Pharmaceuticals and Specialties (25th ed.). Toronto, ON: Webcom. ISBN 0-919115-76-4
- Carlson, NR (2001). Physiology of Behavior-7th ed. Needham Heights, MA: Allyn and Bacon. ISBN 0-205-30840-6
- Gershon, Michael D. (1998). The Second Brain. New York, NY: HarperCollins. ISBN 0-06-018252-0
- Hasselmo, ME (1995). Neuromodulation and cortical function: Modeling the physiological basis of behavior. Behav. Brain Res. 67: 1-27 [2]
- Yu, AJ & Dayan, P (2005). Uncertainty, neuromodulation, and attention. Neuron 46 681-692. [3]
External links
Template:ChemicalSources
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.