Silver
- This page is about silver the chemical element. For the color, see silver (color).
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name, Symbol, Number | silver, Ag, 47 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | transition metals | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 11, 5, d | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | lustrous white metal 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic mass | 107.8682(2) g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Kr] 4d10 5s1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 18, 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (near r.t.) | 10.49 g/cm³ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Liquid density at m.p. | 9.320 g/cm³ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 1234.93 K (961.78 °C, 1763.2 °F) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 2435 K (2162 °C, 3924 °F) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 11.28 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 258 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat capacity | (25 °C) 25.350 J/(mol·K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | cubic face centered | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | 1 (amphoteric oxide) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | 1.93 (Pauling scale) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies | 1st: 731.0 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2nd: 2070 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3rd: 3361 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | 160 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius (calc.) | 165 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 153 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 172 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Miscellaneous | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | diamagnetic | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | (20 °C) 15.87 nΩ·m | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | (300 K) 429 W/(m·K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal diffusivity | (300 K) 174 mm²/s | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | (25 °C) 18.9 µm/(m·K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound (thin rod) | (r.t.) 2680 m/s | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound (thin rod) | (r.t.) 83 m/s | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 30 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 100 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.37 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 2.5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vickers hardness | 251 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 24.5 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS registry number | 7440-22-4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notable isotopes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Silver is a chemical element in the periodic table that has the symbol Ag (from the traditional abbreviation for the Latin Argentum) and atomic number 47. A soft white lustrous transition metal, silver has the highest electrical and thermal conductivity of any metal and occurs in minerals and in free form. This metal is used in coins, jewelry, tableware, and photography.
Notable characteristics
Silver is a very ductile and malleable (slightly harder than gold) univalent coinage metal with a brilliant white metallic luster that can take a high degree of polish. It has the highest electrical conductivity of all metals, even higher than copper, but its greater cost has prevented it from being widely used in place of copper for electrical purposes.
Pure silver also has the highest thermal conductivity, whitest color, the highest optical reflectivity (although it is a poor reflector of ultraviolet), and the lowest contact resistance of any metal. Silver halides are photosensitive and are remarkable for the effect of light upon them. This metal is stable in pure air and water, but does tarnish when it is exposed to ozone, hydrogen sulfide, or air with sulfur in it. The most common oxidation state of silver is +1; a few +2 compounds are known as well.
Applications
The principal use of silver is as a precious metal and its halide salts, especially silver nitrate, are also widely used in photography (which is the largest single end use of silver). Some other uses for silver are as follows:
- Electrical and electronic products, which need silver's superior conductivity, even when tarnished. For example, printed circuits are made using silver paints, and computer keyboards use silver electrical contacts. Silver is also used in high voltage contacts because it is the only metal that will not arc across contacts, hence it is extremely safe.
- Mirrors which need silver's superior reflectivity for visible light are made with silver as the reflecting material in a process called silvering. Common mirrors are backed with aluminium.
- Silver has been coined to produce money since 700 B.C.E. by the Lydians, in the form of electrum. Later, silver was refined and coined in its pure form. The words for "silver" and "money" are the same in at least 14 languages.
- The metal is chosen for its beauty in the manufacture of jewelry and silverware, which are traditionally made from the silver alloy known as Sterling silver, which is 92.5% silver.
- The malleability, non-toxicity and beauty of silver make it useful in dental alloys for fittings and fillings.
- Silver's catalytic properties make it ideal for use as a catalyst in oxidation reactions; for example, the production of formaldehyde from methanol and air by means of silver screens or crystallites containing a minimum 99.95 weight-percent silver.
- Used to make solder and brazing alloys, electrical contacts, and high capacity silver-zinc and silver-cadmium batteries.
- Silver sulfide, also known as Silver Whiskers, is formed when silver electrical contacts are used in an atmosphere rich in hydrogen sulfide.
- Silver fulminate is a powerful explosive.
- Silver chloride can be made transparent and is used as a cement for glass.
- Silver chloride is also a widely used electrode for pH testing and potentiometric measurement
- Silver iodide has been used in attempts to seed clouds to produce rain.
- In legend, silver is traditionally seen as harmful to supernatural creatures like werewolves and vampires. The use of silver fashioned into bullets for firearms is a popular application.
- Silver oxide is used as a positive electrode (cathode) in watch batteries.
- Colloidal silver is an effective antibacterial / antibiotic. It is applied to some bandage materials.
History
Silver (from Anglo-Saxon seolfor, compare Old High German silabar; Ag is from the Latin argentum) has been known since ancient times. It is mentioned in the book of Genesis, and slag heaps found in Asia Minor and on the islands of the Aegean Sea indicate that silver was being separated from lead as early as the 4th millennium B.C.E.
Silver has been used for thousands of years for ornaments and utensils, for trade, and as the basis for many monetary systems. Its value as a precious metal was long considered second only to gold. In Ancient Egypt and Medieval Europe, it was often more valuable than gold.
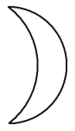
Associated with the moon, as well as with the sea and various lunar goddesses, the metal was referred to by alchemists by the name luna.
One of the alchemical symbols for silver is a crescent moon with the open part on the left (see picture, right).
The metal mercury was thought of as a kind of silver, though the two elements are chemically unrelated; its Latin and English names, hydrargyrum ("watery silver") and quicksilver, respectively, reflect this history.
In heraldry, the argent, in addition to being shown as silver (this has been shown at times with real silver in official representations), can also been shown as white. Occasionally, the word "silver" is used rather than argent; sometimes this is done across-the-board, sometimes to avoid repetition of the word "argent" in blazon.
Europeans found a huge amount of silver in the New World in Zacatecas and Potosí, which triggered a period of inflation in Europe. The conquistador Pizarro was said to have resorted to having his horses shod with silver horseshoes due to the metal's abundance, in contrast to the relative lack of iron in Peru.
The Rio de la Plata was named after silver (in Spanish, plata), and in turn lent the meaning of its name to Argentina.
Occurrence
Silver is found in native form, combined with sulfur, arsenic, antimony, or chlorine and in various ores such as argentite (Ag2S) and horn silver (AgCl). The principal sources of silver are copper, copper-nickel, gold, lead and lead-zinc ores obtained from Canada, Mexico, Peru, Australia and the United States.
This metal is also produced during the electrolytic refining of copper. Commercial grade fine silver is at least 99.9% pure silver and purities greater than 99.999% are available. Mexico is the largest silver producer. According to the Secretary of Economics of Mexico, it produced 80,120,000 troy ounces (2492 metric tons) in 2000, about 15% of the annual production of the world.
Because the lion's share of the world's silver deposits happen to be concentrated in the Americas, silver was far more valuable before the Age of Discovery; on average, about one-sixth or one-seventh the cost of gold. Now, however, silver is relatively cheap compared to other precious metals, and a mass of silver is now worth only about 1/60 the same mass of gold. In turn, copper is about 1/70 as valuable as silver.
Isotopes
Naturally occurring silver is composed of the two stable isotopes Ag-107 and Ag-109 with Ag-107 being the most abundant (51.839% natural abundance). Twenty-eight radioisotopes have been characterized with the most stable being Ag-105 with a half-life of 41.29 days, Ag-111 with a half-life of 7.45 days, and Ag-112 with a half-life of 3.13 hours.
All of the remaining radioactive isotopes have half-lifes that are less than an hour and the majority of these have half lifes that are less than 3 minutes. This element also has numerous meta states with the most stable being Ag-128m (t* 418 years), Ag-110m (t* 249.79 days) and Ag-107m (t* 8.28 days).
Isotopes of silver range in atomic weight from 93.943 u (Ag-94) to 123.929 u (Ag-124). The primary decay mode before the most abundant stable isotope, Ag-107, is electron capture and the primary mode after is beta decay. The primary decay products before Ag-107 are palladium (element 46) isotopes and the primary products after are cadmium (element 48) isotopes.
The palladium isotope Pd-107 decays by beta emission to Ag-107 with a half-life of 6.5 million years. Iron meteorites are the only objects with a high enough Pd/Ag ratio to yield measurable variations in Ag-107 abundance. Radiogenic Ag-107 was first discovered in the Santa Clara meteorite in 1978. The discoverers suggest that the coalescence and differentiation of iron-cored small planets may have occurred 10 million years after a nucleosynthetic event. Pd-107 versus Ag correlations observed in bodies, which have clearly been melted since the accretion of the solar system, must reflect the presence of live short-lived nuclides in the early solar system.
Precautions and health effects
Silver plays no known natural biological role in humans, and possible health effects of silver are a subject of dispute. Silver itself is not toxic but most of its salts are and some may be carcinogenic.
Hippocrates, the father of modern medicine, wrote that silver had beneficial healing and anti-disease properties, and the Phoenicians used to store water, wine, and vinegar in silver bottles to prevent spoiling. The saying 'Born with a silver spoon in the mouth' does not relate to wealth, but to health, as children fed through silver utensils were discovered to be healthier than others (Source: Silver Institute). In the early 1900's people would put silver Dollars in milk bottles to prolong it's freshness. Silver compounds were used sucessfully to prevent infection in World War I before the advent of antibiotics, and Silver compounds are still widely used externally today to accelerate healing in burn victims.
Silver and compounds containing silver (like colloidal silver) can be absorbed into the circulatory system and become deposited in various body tissues leading to a condition called argyria which results in a blue-grayish pigmentation of the skin, eyes, and mucous membranes. Although this condition does not harm a person's health, it is disfiguring and usually permanent. Argyria is rare and mild forms are sometimes mistaken for cyanosis.
Silver-ions and silver compounds show a toxic effect on some bacteria, viruses, algae and fungi typical for heavy metals like lead or mercury, but without the high toxicity to humans that is normally associated with them. It's germicidal effects kills many microbial organisms in vitro (i.e. in a test tube or a petri dish). The exact process by which this is done is still not well understood, although several different theories exist. One of these is a process generally known for heavy metals called the oligodynamic effect, which goes a long way explaining the effect on microbial lifeforms but does not explain certain antiviral functions.
Today, various kinds of silver compounds, or devices to make solutions or colloids containing silver, are sold as remedies for a wide variety of diseases. Although mostly harmless, some people using these home-made solutions use far too much and develop argyria over a period of months or years, and several have been documented in the last few years in the medical literature, including one possible case of coma associated with a high intake of silver (see medical references). It is strongly advised to notify a doctor when taking silver as a form of self-medication.
The widespread use of silver went out of fashion with the invention of antibiotics.
ReferencesISBN links support NWE through referral fees
External links
- WebElements.com – Silver
- Chart: Silver in Dollar
- World mine production of silver, by country
- The Silver Institute A silver industry website
Medical Publications on the effects of silver
- Systemic argyria associated with ingestion of colloidal silver
- Myoclonic status epilepticus following repeated oral ingestion of colloidal silver
- Specific Inactivation of Herpes Simplex Virus by Silver Nitrate at Low Concentrations and Biological Activities of the Inactivated Virus
- Prevention of Herpes Keratoconjunctivitis in Rabbits By Silver Sulfadiazine
ca:Plata cs:Stříbro cy:Arian (elfen) da:Sølv de:Silber et:Hõbe es:Plata eo:Arĝento fr:Argent io:Arjento id:Perak is:Silfur it:Argento he:כסף (יסוד) ku:Zîv lv:Sudrabs lt:Sidabras lb:Sëlwer hu:Ezüst mi:Kawata nl:Zilver nds:Sülver ja:銀 no:Sølv nn:Sølv oc:Argent pl:Srebro pt:Prata ro:Argint ru:Серебро simple:Silver sl:Srebro sr:Сребро fi:Hopea sv:Silver ta:வெள்ளி tt:Kömeş th:เงิน vi:Bạc uk:Срібло zh:銀